Click Chemistry Azide-Alkyne Cycloaddition
In chemical synthesis, "click" chemistry, more commonly called tagging, is a class of biocompatible reactions intended primarily to join substrates of choice with specific biomolecules. Click chemistry is not a single specific reaction, but describes a way of generating products that follow examples in nature, which also generates substances by joining small modular units. In general, click reactions usually join a biomolecule and a reporter molecule. Click chemistry is not limited to biological conditions: the concept of a "click" reaction has been used in pharmacological and various biomimetic applications. However, they have been made notably useful in the detection, localization and qualification of biomolecules.
Click reactions occur in one pot, are not disturbed by water, generate minimal and inoffensive byproducts, and are "spring-loaded"—characterized by a high thermodynamic driving force that drives it quickly and irreversibly to high yield of a single reaction product, with high reaction specificity (in some cases, with both regio- and stereo-specificity). These qualities make click reactions particularly suitable to the problem of isolating and targeting molecules in complex biological environments. In such environments, products accordingly need to be physiologically stable and any byproducts need to be non-toxic (for in vivo systems).
By developing specific and controllable bioorthogonal reactions, scientists have opened up the possibility of hitting particular targets in complex cell lysates. Recently, scientists have adapted click chemistry for use in live cells, for example using small molecule probes that find and attach to their targets by click reactions. Despite challenges of cell permeability, bioorthogonality, background labeling, and reaction efficiency, click reactions have already proven useful in a new generation of pulldown experiments (in which particular targets can be isolated using, for instance, reporter molecules which bind to a certain column), and fluorescence spectrometry (in which the fluorophore is attached to a target of interest and the target quantified or located). More recently, novel methods have been used to incorporate click reaction partners onto and into biomolecules, including the incorporation of unnatural amino acids containing reactive groups into proteins and the modification of nucleotides. These techniques represent a part of the field of chemical biology, in which click chemistry plays a fundamental role by intentionally and specifically coupling modular units to various ends.
The term "click chemistry" was coined by K. Barry Sharpless in 1998, and was first fully described by Sharpless, Hartmuth Kolb, and M.G. Finn of The Scripps Research Institute in 2001.[1][2]
Background[edit]
Click chemistry is a method for attaching a probe or substrate of interest to a specific biomolecule, a process called bioconjugation. The possibility of attaching fluorophores and other reporter molecules has made click chemistry a very powerful tool for identifying, locating, and characterizing both old and new biomolecules.
One of the earliest and most important methods in bioconjugation was to express a reporter on the same open reading frame as a biomolecule of interest. Notably, GFP was first (and still is) expressed in this way at the N- or C- terminus of many proteins. However, this approach comes with several difficulties. For instance, GFP is a very large unit and can often affect the folding of the protein of interest. Moreover, by being expressed at either terminus, the GFP adduct can also affect the targeting and expression of the desired protein. Finally, using this method, GFP can only be attached to proteins, and not post-translationally, leaving other important biomolecular classes (nucleic acids, lipids, carbohydrates, etc.) out of reach.
To overcome these challenges, chemists have opted to proceed by identifying pairs of bioorthogonal reaction partners, thus allowing the use of small exogenous molecules as biomolecular probes. A fluorophore can be attached to one of these probes to give a fluorescence signal upon binding of the reporter molecule to the target—just as GFP fluoresces when it is expressed with the target.
Now limitations emerge from the chemistry of the probe to its target. In order for this technique to be useful in biological systems, click chemistry must run at or near biological conditions, produce little and (ideally) non-toxic byproducts, have (preferably) single and stable products at the same conditions, and proceed quickly to high yield in one pot. Existing reactions, such as Staudinger ligation and the Huisgen 1,3–dipolar cycloaddition, have been modified and optimized for such reaction conditions. Today, research in the field concerns not only understanding and developing new reactions and repurposing and re-understanding known reactions, but also expanding methods used to incorporate reaction partners into living systems, engineering novel reaction partners, and developing applications for bioconjugation.
Reactions[edit]
According to Sharpless, a desirable click chemistry reaction would:[1]
- be modular
- be wide in scope
- give very high chemical yields
- generate only inoffensive byproducts
- be stereospecific
- be physiologically stable
- exhibit a large thermodynamic driving force (> 84 kJ/mol) to favor a reaction with a single reaction product. A distinct exothermic reaction makes a reactant "spring-loaded".
- have high atom economy.
The process would preferably:
- have simple reaction conditions
- use readily available starting materials and reagents
- use no solvent or use a solvent that is benign or easily removed (preferably water)
- provide simple product isolation by non-chromatographic methods (crystallisation or distillation)
Many of the click chemistry criteria are subjective, and even if measurable and objective criteria could be agreed upon, it is unlikely that any reaction will be perfect for every situation and application. However, several reactions have been identified that fit the concept better than others:[clarification needed]
- [3+2] cycloadditions, such as the Huisgen 1,3-dipolar cycloaddition, in particular the Cu(I)-catalyzed stepwise variant,[3] are often referred to simply as Click reactions
- Thiol-ene reaction[4][5]
- Diels-Alder reaction and inverse electron demand Diels-Alder reaction[6]
- [4+1] cycloadditions between isonitriles (isocyanides) and tetrazines[7]
- nucleophilic substitution especially to small strained rings like epoxy [8] and aziridine compounds
- carbonyl-chemistry-like formation of ureas but not reactions of the aldol type due to low thermodynamic driving force.
- addition reactions to carbon-carbon double bonds like dihydroxylation or the alkynes in the thiol-yne reaction.
Copper(I)-Catalyzed Azide-Alkyne Cycloaddition (CuAAC)[edit]
Main article: Azide alkyne Huisgen cycloaddition
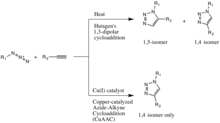
The classic[9][10] click reaction is the Copper-catalyzed reaction of an azide with an alkyne to form a 5-membered heteroatom ring: a Cu(I)-Catalyzed Azide-Alkyne Cycloaddition (CuAAC). The first triazole synthesis, from diethyl acetylenedicarboxylate and phenyl azide, was reported by Arthur Michael in 1893.[11]Later, in the middle of the 20th century, this family of 1,3-dipolar cycloadditions took on Huisgen’s name after his studies of their reaction kinetics and conditions.
The Copper(I)-catalysis of the Huisgen 1,3-dipolar cycloaddition was discovered concurrently and independently by the groups of Valery V. Fokin and K. Barry Sharpless at the Scripps Research Institute in California[12] and Morten Meldal in the Carlsberg Laboratory, Denmark.[13] The copper-catalyzed version of this reaction gives only the 1,4-isomer, whereas Huisgen’s non-catalyzed 1,3-dipolar cycloaddition gives both the 1,4- and 1,5-isomers, is slow, and requires a temperature of 100 degrees Celsius.[11]
Moreover, this copper-catalyzed “click” does not require ligands on the metal, although accelerating ligands such as Tris(triazolyl)methyl amine ligands with various substituents have been reported and used with success in aqueous solution.[11] Other ligands such as PPh3 and TBIA can also be used, even though PPh3 is liable to Staudinger ligation with the azide substituent. Cu2O in water at room temperature was found also to catalyze the same reaction in 15 minutes with 91% yield.[14]
The first reaction mechanism proposed included one catalytic copper atom, but isotope studies have suggested the contribution of two functionally distinct Cu atoms in the CuAAC mechanism.[15]Even though this reaction proceeds effectively at biological conditions, copper in this range of dosage is cytotoxic. Solutions to this problem have been presented, such as using water-soluble ligands on the copper to enhance cell penetration of the catalyst and thereby reduce the dosage needed,[16] or to use chelating ligands to further increase the effective concentration of Cu(I) and thereby decreasing the actual dosage.[17]
Although the Cu(I)-catalyzed variant was first reported by Meldal and co-workers for the synthesis of peptidotriazoles on solid support, they needed more time to discover the full scope of the reaction and were overtaken by the publicly more recognized Sharpless. Meldal and co-workers also chose not to label this reaction type "click chemistry" which allegedly caused their discovery to be largely overlooked by the mainstream chemical society. Sharpless and Fokin independently described it as a reliable catalytic process offering "an unprecedented level of selectivity, reliability, and scope for those organic synthesis endeavors which depend on the creation of covalent links between diverse building blocks."
An analogous RuAAC reaction catalyzed by ruthenium, instead of copper, was reported by the Sharpless group in 2005, and allows for the selective production of 1,5-isomers.[18]
Strain-promoted Azide-Alkyne Cycloaddition (SPAAC)[edit]
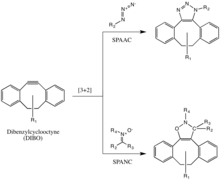
The Bertozzi group further developed one of Huisgen’s copper-free click reactions to overcome the cytotoxicity of the CuAAC reaction.[19] Instead of using Cu(I) to activate the alkyne, the alkyne is instead introduced in a strained difluorooctyne (DIFO), in which the electron-withdrawing, propargylic, gem-fluorines act together with the ring strain to greatly destabilize the alkyne.[20] This destabilization increasing the reaction driving force, and the desire of the cycloalkyne to relieve its ring strain.
This reaction proceeds as a concerted [3+2] cycloaddition in the same mechanism as the Huisgen 1,3-dipolar cycloaddition. Substituents other than fluorines, such as benzene rings, are also allowed on the cyclooctyne.
This reaction has been used successfully to probe for azides in living systems, even though the reaction rate is somewhat slower than that of the CuAAC. Moreover, because the synthesis of cyclooctynes often gives low yield, probe development for this reaction has not been as rapid as for other reactions. But cyclooctyne derivatives such as DIFO, dibenzylcyclooctyne (DIBO) and biarylazacyclooctynone (BARAC) have all been used successfully in the SPAAC reaction to probe for azides in living systems.[21][22][23]
Strain-promoted Alkyne-Nitrone Cycloaddition (SPANC)[edit]
Diaryl-strained-cyclooctynes including dibenzylcyclooctyne (DIBO) have also been used to react with 1,3-nitrones in strain-promoted alkyne-nitrone cycloadditions (SPANC) to yield N-alkylated isoxazolines.[24]
Because this reaction is metal-free and proceeds with fast kinetics (k2 as fast as 60 1/Ms, faster than both the CuAAC or the SPAAC) SPANC can be used for live cell labeling. Moreover, substitution on both the carbon and nitrogen atoms of the nitrone dipole, and acyclic and endocyclic nitrones are all tolerated. This large allowance provides a lot of flexibility for nitrone handle or probe incorporation.[25]
However, the isoazoline product is not as stable as the triazole product of the CuAAC and the SpAAC, and can undergo rearrangements at biological conditions. Regardless, this reaction is still very useful as it has notably fast reaction kinetics.[24]
The applications of this reaction include labeling proteins containing serine as the first residue: the serine is oxidized to aldehyde with NaIO4 and then converted to nitrone with p-methoxybenzenethiol, N-methylhydroxylamine and p-ansidine, and finally incubated with Cyclooctyne to give a click product. The SPANC also allows for multiplex labeling.[26][27]
Reactions of Strained Alkenes[edit]
Strained alkenes also utilize strain-relief as a driving force that allows for their participation in click reactions. Trans-cycloalkenes (usually cyclooctenes) and other strained alkenes such as oxanorbornadiene react in click reactions with a number of partners including Azides, Tetrazines and Tetrazoles. These reaction partners can interact specifically with the strained alkene, staying bioorthogonal to endogenous alkenes found in lipids, fatty acids, cofactors and other natural products.[26]
Alkene and Azide [3+2] cycloaddition[edit]
Oxanorbornadiene (or another activated alkene) reacts with azides, giving triazoles as a product. However, these product triazoles are not aromatic as they are in the CuAAC or SPAAC reactions, and as a result are not as stable. The activated double bond in oxanobornadiene makes a triazoline intermediate that subsequently spontaneously undergoes a retro Diels-alder reaction to release furan and give 1,2,3- or 1,4,5-triazoles. Even though this reaction is slow, it is useful because oxabornodiene is relatively simple to synthesize. The reaction is not, however, entirely chemoselective.[28]
Alkene and Tetrazine inverse-demand Diels-Alder[edit]
Strained cyclooctenes and other activated alkenes react with tetrazines in an inverse electron-demand Diels-Alder followed by a retro [4+2] cycloaddition (see figure).[29] Like the other reactions of the trans-cyclooctene, ring strain release is a driving force for this reaction. Thus, three-membered and four-membered cycloalkenes, due to their high ring strain, make ideal alkene substrates.[29]
Similar to other [4+2] cycloadditions, electron-donating substituents on the dienophile and electron-withdrawing substituents on the diene accelerate the inverse-demand diels-alder. The diene, the tetrazine, by virtue of having the additional nitrogens, is a good diene for this reaction. The dienophile, the activated alkene, can often be attached to electron-donating alkyl groups on target molecules, thus making the dienophile more suitable for the reaction.[30]
Alkene and Tetrazole photoclick reaction[edit]
The Tetrazole-alkene “photoclick” reaction is another dipolar addition that Husigen first introduced about 50 years ago (ChemBioChem 2007, 8, 1504. (68) Clovis, J. S.; Eckell, A.; Huisgen, R.; Sustmann, R. Chem. Ber. 1967, 100, 60.) Tetrazoles with amino or styryl groups that can be activated by UV light at 365 nm (365 does not damage cells) react quickly (so that the UV light does not have to be on for a long time, usually around 1–4 minutes) to make fluorogenic pyrazoline products. This reaction scheme is well suited for the purpose of labeling in live cells, because UV light at 365 nm damages cells minimally. Moreover, the reaction proceeds quickly, so that the UV light can be administered for short durations. Finally, the non-fluorogenic reactants give rise to a fluorogenic product, equipping the reaction with a built-in spectrometry handle.
Both tetrazoles and the alkene groups have been incorporated as protein handles as unnatural amino acids, but this benefit is not unique. Instead, the photoinducibility of the reaction makes it a prime candidate for spatiotemporal specificity in living systems. Challenges include the presence of endogenous alkenes, though usually cis (as in fatty acids) they can still react with the activated tetrazole.[31]
Applications[edit]
The applications of click chemistry are broad in that they allow for the attachment of a wide range of probes to a wide range of biomolecule targets. Several notable applications of click chemistry include the attachment of fluorescent probes for spectrometric quantification and qualification, and of handle molecules that allow for purification of the target biomolecule. For example, fluorophores such as rhodamine have been coupled onto norbonene, and reacted with tetrazine in living systems.[32] In other cases, SPAAC between a cyclooctyne-modified fluorophore and azide-tagged proteins allowed the selection of these proteins in cell lysates.[33]
In addition, novel methods for the incorporation of click reaction partners into systems in and ex vivo contribute to the scope of possible reactions. The development of unnatural amino acid incorporation by ribosomes has allowed for the incorporation of click reaction partners as unnatural side groups on these unnatural amino acids. For example, an UAA with an azide side group provides convenient access for cycloalkynes to proteins tagged with this “AHA” unnatural amino acid.[34] In another example, “CpK” has a side group including a cyclopropane alpha to an amide bond that serves as a reaction partner to tetrazine in an inverse diels-alder reaction.[35]
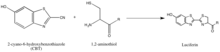
The synthesis of luciferin exemplifies another strategy of isolating reaction partners, which is to take advantage of rarely-occurring, natural groups such as the 1,2-aminothiol, which appears only when a cysteine is the final N’ amino acid in a protein. Their natural selectivity and relative bioorthogonality is thus valuable in developing probes specific for these tags. The above reaction occurs between a 1,2-aminothiol and a 2-cyanobenzothiazole to make luciferin, which is fluorescent. This luciferin fluorescence can be then quantified by spectrometry following a wash, and used to determine the relative presence of the molecule bearing the 1,2-aminothiol. If the quantification of non-1,2-aminothiol-bearing protein is desired, the protein of interest can be cleaved to yield a fragment with a N’ Cys that is vulnerable to the 2-CBT.[36]
Additional applications include:
- Two-dimensional gel electrophoresis separation[37]
- preparative organic synthesis of 1,4-substituted triazoles
- modification of peptide function with triazoles
- modification of natural products and pharmaceuticals
- natural product discovery [38]
- drug discovery
- macrocyclizations using Cu(I) catalyzed triazole couplings
- modification of DNA and nucleotides by triazole ligation
- supramolecular chemistry: calixarenes, rotaxanes, and catenanes
- dendrimer design
- carbohydrate clusters and carbohydrate conjugation by Cu(1) catalyzed triazole ligation reactions
- Polymers and Biopolymers [39]
- surfaces[40]
- material science
- nanotechnology,[41] and
- Bioconjugation, for example, azidocoumarin.
- Biomaterials[42]
In combination with combinatorial chemistry, high-throughput screening, and building chemical libraries, click chemistry has sped up new drug discoveries by making each reaction in a multistep synthesis fast, efficient, and predictable.
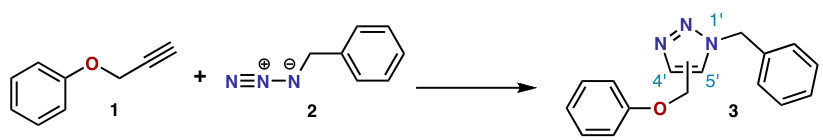
The Azide-Alkyne Huisgen Cycloaddition is a 1,3-dipolar cycloaddition between an azide and a terminal or internal alkyne to give a 1,2,3-triazole. Rolf Huisgen[1] was the first to understand the scope of this organic reaction. American chemist K. Barry Sharpless has referred to this cycloaddition as "the cream of the crop" of click chemistry[2] and "the premier example of a click reaction."[3]
In the reaction above[4] azide 2 reacts neatly with alkyne 1 to afford the triazole 3 as a mixture of 1,4-adduct and 1,5-adduct at 98 °C in 18 hours.
The standard 1,3-cycloaddition between an azide 1,3-dipole and an alkene as dipolarophile has largely been ignored due to lack of reactivity as a result of electron-poor olefins and elimination side reactions. Some success has been found with non-metal-catalyzed cycloadditions, such as the reactions using dipolarophiles that are electron-poor olefins[5] or alkynes.
Although azides are not the most reactive 1,3-dipole available for reaction, they are preferred for their relative lack of side reactions and stability in typical synthetic conditions.
Copper catalysis
A notable variant of the Huisgen 1,3-dipolar cycloaddition is the copper(I) catalyzed variant, no longer a true concerted cycloaddition, in which organic azides and terminal alkynes are united to afford 1,4-regioisomers of 1,2,3-triazoles as sole products (substitution at positions 1' and 4' as shown above). The copper(I)-catalyzed variant was first reported in 2002 in independent publications by Morten Meldal at the Carlsberg Laboratory in Denmark[6] and Valery Fokin and K. Barry Sharpless at the Scripps Research Institute.[7]While the copper(I)-catalyzed variant gives rise to a triazole from a terminal alkyne and an azide, formally it is not a 1,3-dipolar cycloaddition and thus should not be termed a Huisgen cycloaddition. This reaction is better termed the Copper(I)-catalyzed Azide-Alkyne Cycloaddition (CuAAC).
While the reaction can be performed using commercial sources of copper(I) such as cuprous bromide or iodide, the reaction works much better using a mixture of copper(II) (e.g. copper(II) sulfate) and a reducing agent (e.g. sodium ascorbate) to produce Cu(I) in situ. As Cu(I) is unstable in aqueous solvents, stabilizing ligands are effective for improving the reaction outcome, especially if tris-(benzyltriazolylmethyl)amine (TBTA) is used. The reaction can be run in a variety of solvents, and mixtures of water and a variety of (partially) miscible organic solvents including alcohols, DMSO, DMF, tBuOH and acetone. Owing to the powerful coordinating ability of nitriles towards Cu(I), it is best to avoid acetonitrile as the solvent. The starting reagents need not be completely soluble for the reaction to be successful. In many cases, the product can simply be filtered from the solution as the only purification step required.
NH-1,2,3-triazoles are also prepared from alkynes in a sequence called the Banert cascade.
The utility of the Cu(I)-catalyzed click reaction has also been demonstrated in the polymerization reaction of a bis-azide and a bis-alkyne with copper(I) and TBTA to a conjugated fluorene based polymer.[8] The degree of polymerization easily exceeds 50. With a stopper molecule such as phenyl azide, well-defined phenyl end-groups are obtained.
The copper-mediated azide-alkyne cycloaddition is receiving widespread use in material and surface sciences.[9] Most variations in coupling polymers with other polymers or small molecules have been explored. Current shortcomings are that the terminal alkyne appears to participate in free radical polymerizations. This requires protection of the terminal alkyne with a trimethyl silyl protecting group and subsequent deprotection after the radical reaction are completed. Similarly the use of organic solvents, copper (I) and inert atmospheres to do the cycloaddition with many polymers makes the "click" label inappropriate for such reactions. An aqueous protocol for performing the cycloaddition with free radical polymers is highly desirable.
The CuAAC click reaction also effectively couples polystyrene and bovine serum albumin (BSA).[10] The result is an amphiphilic biohybrid. BSA contains a thiol group at Cys-34 which is functionalized with an alkyne group. In water the biohybrid micelles with a diameter of 30 to 70 nanometer form aggregates.
Copper catalysts
The use of a Cu catalyst in water was an improvement over the same reaction first popularized by Rolf Huisgen in the 1970s, which he ran at elevated temperatures.[11] The traditional reaction is slow and thus requires high temperatures. However, the azides and alkynes are both kinetically stable.
As mentioned above, copper-catalysed click reactions work essentially on terminal alkynes. The Cu species undergo metal insertion reaction into the terminal alkynes. The Cu(I) species may either be introduced as preformed complexes, or are otherwise generated in the reaction pot itself by one of the following ways:
- A Cu compound (in which copper is present in the +2 oxidation state) is added to the reaction in presence of a reducing agent (e.g. sodium ascorbate) which reduces the Cu from the (+2) to the (+1) oxidation state. The advantage of generating the Cu(I) species in this manner is it eliminates the need of a base in the reaction. Also the presence of reducing agent makes up for any oxygen which may have gotten into the system. Oxygen oxidises the Cu(I) to Cu(II) which impedes the reaction and results in low yields. One of the more commonly used Cu compounds is CuSO4
- Oxidation of Cu(0) metal
- Halides of copper may be used where solubility is an issue. However, the iodide and bromide Cu salts require either the presence of amines or higher temperatures.
Commonly used solvents are polar aprotic solvents such as THF, DMSO, Acetonitrile, DMF as well as in non-polar aprotic solvents such as toluene. Neat solvents or a mixture of solvents may be used.
DIPEA (N,N-Diisopropylethylamine) and Et3N (triethylamine) are commonly used bases.[12]
Mechanism
A mechanism for the reaction has been suggested based on density functional theory calculations.[13]Copper is a 1st row transition metal. It has the electronic configuration [Ar] 3d10 4s1. The copper (I) species generated in situ forms a pi complex with the triple bond of a terminal alkyne. In the presence of a base, the terminal hydrogen, being the most acidic is deprotonated first to give a Cu acetylide intermediate. Studies have shown that the reaction is second order with respect to Cu. It has been suggested that the transition state involves two copper atoms. One copper atom is bonded to the acetylide while the other Cu atom serves to activate the azide. The metal center coordinates with the electrons on the nitrogen atom. The azide and the acetylide are not coordinated to the same Cu atom in this case. The ligands employed are labile and are weakly coordinating. The azide displaces one ligand to generate a copper-azide-acetylide complex. At this point cyclisation takes place. This is followed by protonation; the source of proton being the hydrogen which was pulled off from the terminal acetylene by the base. The product is formed by dissociation and the catalyst ligand complex is regenerated for further reaction cycles.
The reaction is assisted by the copper, which, when coordinated with the acetylide lowers the pKa of the alkyne C-H by up to 9.8 units. Thus under certain conditions, the reaction may be carried out even in the absence of a base.
In the uncatalysed reaction the alkyne remains a poor electrophile. Thus high energy barriers lead to slow reaction rates.[14]
Ligand assistance
The ligands employed are usually labile i.e. they can be displaced easily. Though the ligand plays no direct role in the reaction the presence of a ligand has its advantages. The ligand protects the Cu ion from interactions leading to degradation and formation of side products and also prevents the oxidation of the Cu(I) species to the Cu(II). Furthermore, the ligand functions as a proton acceptor thus eliminating the need of a base.[15]
Ruthenium catalysis
The ruthenium-catalysed 1,3-dipolar azide-alkyne cycloaddition (RuAAC) gives the 1,5-triazole. Unlike CuAAC in which only terminal alkynes reacted, in RuAAC both terminal and internal alkynes can participate in the reaction. This suggests that ruthenium acetylides are not involved in the catalytic cycle.
The proposed mechanism suggests that in the first step, the spectator ligands undergo displacement reaction to produce an activated complex which is converted, via oxidative coupling of an alkyne and an azide to the ruthenium containing metallocyle (Ruthenacycle). The new C-N bond is formed between the more electronegative and less sterically demanding carbon of the alkyne and the terminal nitrogen of the azide. The metallacycle intermediate then undergoes reductive elimination releasing the aromatic triazole product and regenerating the catalyst or the activated complex for further reaction cycles.
Cp*RuCl(PPh3)2, Cp*Ru(COD)and Cp*[RuCl4] are commonly used ruthenium catalysts. Catalysts containing cyclopentadienyl(Cp) group are also used. However, better results are observed with the pentamethylcyclopentadienyl(Cp*) version. This may be due to the sterically demanding Cp* group which facilitates the displacement of the spectator ligands.[16][17]
Silver catalysis
Recently, the discovery of a general Ag(I)-catalyzed azide–alkyne cycloaddition reaction (Ag-AAC) leading to 1,4-triazoles is reported. Mechanistic features are similar to the generally accepted mechanism of the copper(I)-catalyzed process. Interestingly, silver(I)-salts alone are not sufficient to promote the cycloaddition. However the ligated Ag(I) source has proven to be exceptional for AgAAC reaction.[18] [19]Curiously, pre-formed silver acetylides do not react with azides; however, silver acetylides do react with azides under catalysis with copper(I).[20]
Recent Literature

A Novel Approach to 1-Monosubstituted 1,2,3-Triazoles by a Click Cycloaddition/Decarboxylation Process
M. Xu, C. Kuang, Z. Wang, Q. Yang, Y. Jiang, Synthesis, 2011, 223-228.

The Use of Calcium Carbide in the Synthesis of 1-Monosubstituted Aryl 1,2,3-Triazole via Click Chemistry
Y. Jiang, C. Kuang, Q. Yang, Synlett, 2009, 3163-3166.

Microwave Irradiation as an Effective Means of Synthesizing Unsubstituted N-Linked 1,2,3-Triazoles from Vinyl Acetate and Azides
S. G. Hansen, H. H. Jensen, Synlett, 2009, 3275-3278.

A Convenient Synthesis of 1-Substituted 1,2,3-Triazoles via CuI/Et3N Catalyzed ‘Click Chemistry' from Azides and Acetylene Gas
L.-Y. Wu, Y.-X. Xie, Z.-S. Chen, Y.-N. Niu, Y.-M. Liang, Synlett, 2009, 1453-1456.

2-Ethynylpyridine-Promoted Rapid Copper(I) Chloride Catalyzed Azide-Alkyne Cycloaddition Reaction in Water
H. Hiroki, K. Ogata, S.-i. Fukuzawa, Synlett, 2013, 24, 843-846.

A New Simplified Protocol for Copper(I) Alkyne-Azide Cycloaddition Reactions Using Low Substoichiometric Amounts of Copper(II) Precatalysts in Methanol
B. R. Buckley, M. M. P. Figueres, A. N. Khan, H. Heaney, Synlett, 2016, 27, 51-56.

Tandem Catalysis: From Alkynoic Acids and Aryl Iodides to 1,2,3-Triazoles in One Pot
A. Kolarovič, M. Schnürch, M. D. Mihovilovic, J. Org. Chem., 2011, 76, 2613-2618.

Self-assembly of copper sulfate and a poly(imidazole-acrylamide) amphiphile provides a highly active, reusable, globular, solid-phase catalyst for click chemistry. The insoluble amphiphilic polymeric imidazole Cu catalyst drove the cycloaddition of various of alkynes and organic azides at very low catalyst loadings and can be readily reused without loss of activity to give the corresponding triazoles quantitatively.
Y. M. A. Yamada, S. M. Sarkar, Y. Uozumi, J. Am. Chem. Soc., 2012, 134, 9285-9286.

Copper-Catalyzed Azide-Alkyne Cycloaddition Reaction in Water Using Cyclodextrin as a Phase Transfer Catalyst
J.-A. Shin, Y.-G. Lim, K.-H. Lee, J. Org. Chem., 2012, 77, 4117-4122.

In the presence of a readily accessible nanosized Cu/Fe bimetallic system, Cu-catalyzed azide-alkyne cycloadditions can easily be achieved at ambient temperature with high efficiency. The catalyst produces significantly lower copper contaminants compared to homogeneous copper complexes. Iron not only behaves as support, but also acts as a redox scavenger that reduces the copper contamination of the organic product.
S. Kovács, K. Zih-Peréni, Á. Révész, Z. Novák, Synthesis, 2012, 44, 3722-3730.

CuI/DIPEA/HOAc is as a highly efficient catalytic system for CuAAC. In this novel acid-base jointly promoted formation of 1,2,3-triazoles, HOAc was recognized to accelerate the conversions of the C-Cu bond-containing intermediates and buffer the basicity of DIPEA. As a result, all drawbacks occurring in the popular catalytic system CuI/NR3 were overcome easily.
C. Shao, X. Wang, Q. Zhang, S. Luo, J. Zhao, Y. Hu, J. Org. Chem., 2011, 76, 6832-6836.

[CuBr(PPh3)3] for Azide-Alkyne Cycloaddition Reactions under Strict Click Conditions
S. Lal, S. Díez-González, J. Org. Chem., 2011, 76, 2367-2373.

A Copper(I) Isonitrile Complex as a Heterogeneous Catalyst for Azide-Alkyne Cycloaddition in Water
M. Liu, O. Reiser, Org. Lett., 2011, 13, 1102-1105.

An abnormal NHC complex of copper with 1,4-diphenyl-1,2,3-triazol-5-ylidene [CuCl(TPh)] efficiently catalyzed click reactions of azides with alkynes to give 1,4-substituted 1,2,3-triazoles in excellent yields at room temperature with short reaction times. CuCl(TPh) was particularly effective for the reaction between sterically hindered azides and alkynes.
T. Nakamura, T. Terashima, K. Ogata, S.-i. Fukuzawa, Org. Lett., 2011, 13, 620-623.

Carboxylic Acid-Promoted Copper(I)-Catalyzed Azide-Alkyne Cycloaddition
C. Shao, X. Wang, J. Xu, J. Zhao, Q. Zhang, Y. Hu, J. Org. Chem., 2010, 75, 7002-7005.

A Highly Active Catalyst for Huisgen 1,3-Dipolar Cycloadditions Based on the Tris(triazolyl)methanol-Cu(I) Structure
S. Özçubukçu, E. Ozkal, C. Jimeno, M. A. Pericàs, Org. Lett., 2009, 11, 4680-4683.

Benzimidazole and Related Ligands for Cu-Catalyzed Azide-Alkyne Cycloaddition
V. O. Rodionov, S. I. Presolski, S. Gardinier, Y.-H. Lim, M. G. Finn, J. Am. Chem. Soc., 2007, 129, 12696-12704.

1,3-Dipolar Cycloaddition of Organic Azides to Alkynes by a Dicopper-Substituted Silicotungstate
K. Kamata, Y. Nakagawa, K. Yamaguchi, N. Mizuno, J. Am. Chem. Soc., 2008, 130, 15304-15310.

Heterogeneous Copper Catalyst for the Cycloaddition of Azides and Alkynes without Additives under Ambient Conditions
I. S. Park, M. S. Kwon, Y. Kim, J. S. Lee, J. Park, Org. Lett., 2008, 10, 497-500.

Heterogeneous Copper-in-Charcoal-Catalyzed Click Chemistry
B. H. Lipshutz, B. R. Taft, Angew. Chem. Int. Ed., 2006, 45, 8235-8238.

A Stepwise Huisgen Cycloaddition Process: Copper(I)-Catalyzed Regioselective Ligation of Azides and Terminal Alkynes
V. V. Rostovtsev, L. G. Green, V. V. Fokin, K. B. Sharpless, Angew. Chem. Int. Ed., 2002, 41, 2596-2599.

Regioselective Synthesis of 1,2,3-Triazoles by Use of a Silica-Supported Copper(I) Catalyst
T. Miaoa, L. Wang, Synthesis, 2008, 363-368.

Efficient Conversion of Aromatic Amines into Azides: A One-Pot Synthesis of Triazole Linkages
K. Barral, A. D. Moorhouse, J. E. Moses, Org. Lett., 2007, 9, 1809-1811.

One-Pot Three-Step Synthesis of 1,2,3-Triazoles by Copper-Catalyzed Cycloaddition of Azides with Alkynes formed by a Sonogashira Cross-Coupling and Desilylation
F. Friscourt, G.-J. Boons, Org. Lett., 2010, 12, 4936-4939.

Copper(II)-Catalyzed Conversion of Aryl/Heteroaryl Boronic Acids, Boronates, and Trifluoroborates into the Corresponding Azides: Substrate Scope and Limitations
K. D. Grimes, A. Gupte, C. C. Aldrich, Synthesis, 2010, 1441-1448.

Ruthenium-Catalyzed Cycloaddition of Aryl Azides and Alkynes
L. K. Rasmussen, B. C. Boren, V. V. Fokin, Org. Lett., 2007, 9, 5337-5339.

Efficient one-pot synthesis of polysubstituted 6-[(1H-1,2,3-triazol-1-yl)methyl]uracils through the "click" protocol
P. Jansa, P. Špaček, I. Votruba, P. Břehová, M. Dračínský, B. Klepetářová, Z. Janeba, Collect. Czech. Chem. Commun., 2011, 13, 1121-1131.






Iam very happy to have such a blog here.I searched a lot and at last i find out the apt one..This blog is very informative for me and your knowledge about this topic is appreciable . Sohman Epoxy also contains a lot of information.This website also help me to deepen my knowledge..Just check this one also Benzyl Alcohol surely you may get some more details about the topic.Thank you.
ReplyDelete